Introduction
Most fractures heal in a predictable timeline, relative to the bone that is injured, within several months from the time of injury.1 Treatment of fractures ranges from non-surgical management involving immobilization strategies to surgical management using fixation methods.2,3 Research has shown that normal bone healing undergoes three phases: inflammation, repair, and remodeling.4 However, approximately 100,000 fractures in the United States (US) each year result in the ABH (Adverse Bone Healing) event.1 ABH event includes fracture non-union, delayed union, or malunion, which is problematic due to a prolonged recovery process involving continued pain or dysfunction, increased risk for surgical intervention, and extended periods of disability. Thus, efforts are focused on improving bone healing through pharmacological, surgical, cellular, and biophysical approaches.5
Fractures are associated with varied levels of pain that must be managed by the clinical care team with the use of a structured pain regimen. Most adult trauma patients in the US receive prescriptions for opioids due to high pain levels associated with trauma.6 While opioids are known to be powerful analgesics, their side effects and addictive potential have driven the need for alternative approaches to pain management.7 Of the known alternatives, non-steroidal anti-inflammatory drugs (NSAIDs) are of particular interest because studies have found that they can be as efficacious in managing pain as opioids with less addictive potential.8–10 However, the use of NSAIDs in orthopedic trauma patients is controversial due to previous studies reporting the possible association of NSAID use with delayed union and nonunion.
Bone healing after a fracture is supported by a prostaglandin-mediated inflammatory response dependent on cyclo-oxygenase (COX) enzymes.11,12 NSAIDs exhibit their anti-inflammatory and analgesic effects through inhibition of COX enzymes.13 By hindering the enzyme involved in both pain and healing, NSAID use is thought to contribute to ABH events. The hindrance of bone healing by NSAID use has been well documented in animal studies. Still, the clinical significance in humans is unclear, and various factors may modify the relationship.14 A retrospective study looking at human long-bone fractures has reported that NSAID use increased the risk of non-union or malunion by a factor of two compared to patients who were not exposed to NSAIDs after the injury.15 In contrast, a prospective study examining NSAID use in spinal fusions demonstrated that short-term NSAID use did not hinder bone healing. Still, prolonged exposure (>6 weeks) increased the risk of nonunion.16
A meta-analysis, including literature between 1966 and 2008, found that an increased risk of non-union following NSAID use was detected, but the effect was lost when only higher-quality studies were included in the analysis.17 A more recent meta-analysis using literature between 1990 and 2016 reported that the use of NSAIDs doubles the risk of adverse bone healing compared to patients who are not exposed to NSAIDs.18 However, this analysis compiled fractures, arthrodesis, and osteotomies into a single analysis, which limits generalizability and clinical utility. To our knowledge, the most recent meta-analysis that examined the effect of NSAID use on fracture healing exclusively included high-quality Randomized Controlled Trials (RCTs) up to October 2018 and reported a 3.47 increase in odds of non-union following the use of NSAIDs.19
This project aims to extend the current state of literature by providing an updated meta-analysis on the effect of NSAID use, specifically on fracture healing. It will encompass a variety of study designs, including RCTs, cohort studies, case-control studies, and case series. The current study will not include studies on patients undergoing arthrodesis, given that fusion healing represents a different healing process.20–23 The study hypothesis is that prescribing NSAIDs following fracture treatment will not cause an increase in ABH events.
Methods
Databases/Literature Search
A search of literature was conducted through computerized databases. MEDLINE (PubMed and Ovid), Embase, Cochrane Database of Systematic Reviews, and Web of Science were searched for relevant articles. Keywords and corresponding Medical Subject Heading (MeSH) terms for the search were identified according to prior literature and modified to best fit our study purpose. Appendix A lists the final search terms and the search strategy used for this study. Literature published between January 1st, 1990, and June 1st, 2023, was included in the search. Given that the study was a meta-analysis, an Institutional Review Board review was not applicable.
Study Selections
After compiling articles from the initial database search, two authors (JTS and ER) conducted a sequential review of titles, abstracts, and full articles to determine inclusion in the final analysis. In the three sequential rounds of reviews, each of the two authors independently determined eligibility for inclusion. Articles were included if either of the two reviewers determined the article eligible for the next round. In the full article review, discrepancies in the determination of eligibility were brought to a third author (SR) and discussed until a consensus was reached. After the full article review, study quality was evaluated using the Newcastle-Ottawa Scale for case-control and cohort studies and the Cochrane Collaboration Risk of Bias Assessment Tool for RCTs.24,25 Studies were removed from the final analysis with a score of six and above for Cochrane Collaboration’s assessment tool and 5 and below for the Newcastle-Ottawa scale.
Inclusion and Exclusion Criteria
Studies of pediatric and adult patients with any fracture type, which examined the non-union and/or delayed union rates between patients exposed to NSAIDs versus patients not exposed to NSAIDs published between January 1st, 1990, and June 1st, 2023, were included in the analysis. Studies examining short-term peri-operative exposure to NSAIDs were also included in the analysis. RCTs, cohort studies, case-control studies, and case series with ≥ 5 patients were included. Radiologic determination of adverse bone healing at a minimum of six months follow-up or an adverse bone healing event noted in electronic medical records following the fracture was the basis of the outcome of interest. Studies with zero ABH events in both NSAID-exposed and non-exposed groups were also excluded.
Data
The primary outcome of interest was a composite rate of union, delayed union, and non-union after fractures. The primary exposure of interest was whether the patient with the fracture was exposed to NSAIDs during the process of fracture healing. From each article included in the final analysis, the number of subjects pertaining to the following four groups were extracted: exposed to NSAIDs with timely healing, exposed to NSAIDs with adverse healing, not exposed to NSAIDs with timely healing, and not exposed to NSAIDs with adverse healing. Additionally, subjects’ ages, study design, and operative techniques involved in fracture care were extracted from each publication.
Statistical Analysis
The statistical analysis was completed per QUORUM (Quality of Reporting of Meta-analyses) and PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines for meta-analyses.26,27 A maximum likelihood random-effects model was used to minimize the variance among the included studies. A meta-regression was completed on the pooled primary outcome and expressed as odds ratios with 95% confidence intervals. Odds Ratios for each study, as well as for the cumulative analysis, were calculated. Separate subgroup analysis was completed by stratifying publications that included pediatric versus adult patients. The statistical significance of the meta-regression analysis was evaluated using the Wald-type test with an alpha of 0.05 for all analyses.
Results
The keywords search from OVID, Cochrane, and Embase databases yielded 3,364, 136, and 365 articles, respectively, for a total of 3,865 articles with 889 duplicates [Figure 1]. A total of 3,050 were screened for their titles, 471 for their abstracts, and 108 for their full-text articles, which yielded eight articles for inclusion. The chance-adjusted kappa scores between the two authors (JTS & ER) for the title, abstract, and full-text screens were 0.71, 0.78, and 0.84, respectively. In a process known as reference snowballing,28 a manual search of references from the 108 full-text article screening yielded four additional articles to be included in the final analysis.
Included studies were published between 2000 and 2020 [Table 1]. All studies included from the screening process, and the reference snowballing satisfied the criteria for study quality assessments. Four of the twelve included studies were RCTs, and eight were retrospective. The pooled analysis included a total of 645,891 patients, of which 584,848 were not exposed to NSAIDs, and 61,043 were exposed to NSAIDs at the time or after the time of fracture. A total of 21,785 (of which 16,908 were not exposed to NSAIDs and 4,877 were exposed to NSAIDs) cases of ABH events were included in the analysis. The proportion of patients who were determined to have ABH events was variable both within patients who were not exposed to NSAIDs (Range: 0.8% - 19.0%) and within patients who were exposed to NSAIDs (Range: 0.6% – 69.0%).
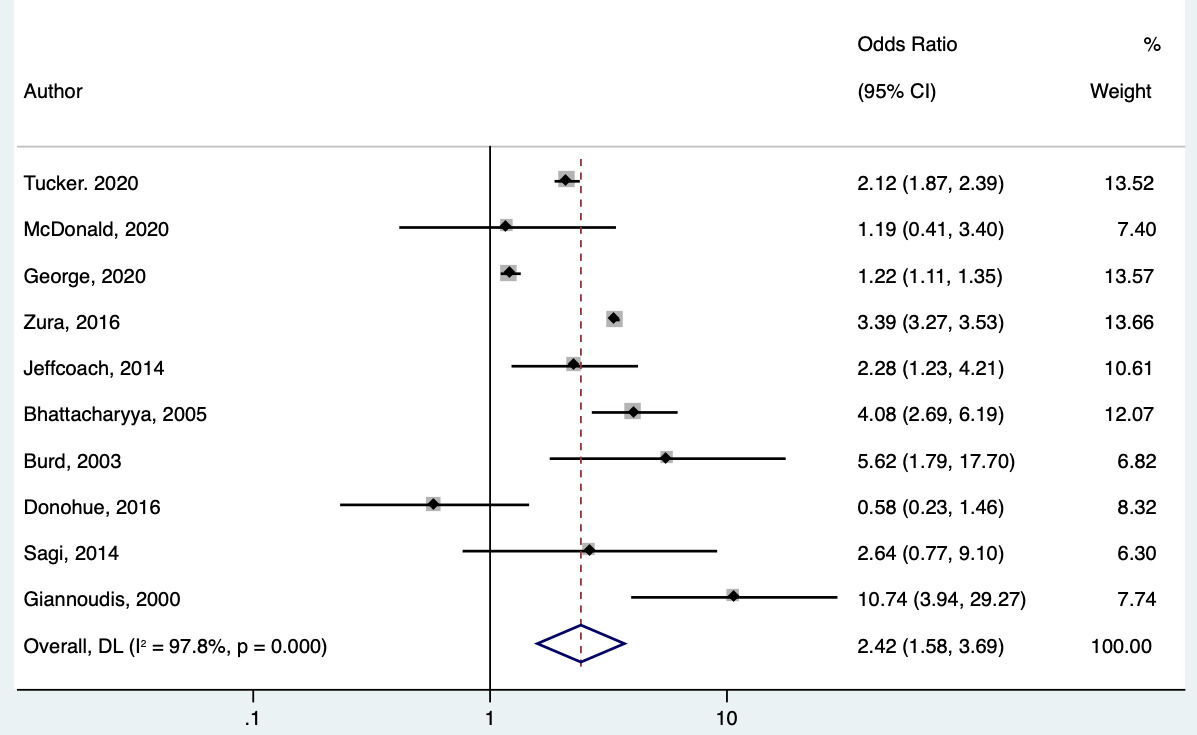
The pooled analysis of 12 included studies resulted in an Odds Ratio of 2.10 for ABH events using NSAIDs (95% CI: 1.41 - 3.15) [Figure 2]. The odds ratio for ABH events with use of NSAIDs among the 12 studies ranged between 0.58 and 10.74. When stratified by age, the odds ratio of ABH events with the use of NSAIDs was 2.41 (95% CI: 1.58-3.69) among adults [Figure 3] and 0.741 (95% CI: 0.351-1.565) among pediatric patients [Figure 4].
Discussion
The cumulative analysis of current literature suggests that patients who are exposed to NSAIDs post-fracture are twice as likely to experience ABH events compared to patients who are not exposed to NSAIDs post-fracture. This is specifically relevant among adult patients, given that patients ≥ 18 years old yielded an odds ratio approaching two and a half times increased likelihood of ABH events. In contrast, pediatric patients did not show a significant change in the prevalence of ABH events when exposed to NSAIDs. However, the wide range of odds ratios among the included studies may indicate several confounding variables that could potentially affect the association between NSAID use and ABH events. Specifically, this meta-analysis findings should be interpreted with the understanding that the type, dose, and duration of NSAID treatment were not controlled for. Moreover, each bone has its own local and vascular dynamics, resulting in distinct risks of ABH events that may make NSAID use applicable in some cases and not others. Future studies should focus on definitive guidelines for the safe usage of NSAIDs, specifically tailored to different patient populations and fracture characteristics.
This study’s findings are consistent with the meta-analysis performed by Wheatley et al., which included all study types as well as both arthrodesis and fractures.18 Six of the included studies overlapped with the analysis in this study, while six additional studies on fracture healing were included in the analysis in this study. Whereas Wheatley et al. (2018) examined both arthrodesis and fracture outcomes following NSAID use, this study examined fracture outcomes exclusively.18 It is noteworthy that similar odds ratios were reported in both studies. The finding that post-fracture NSAID exposure did not show an increased likelihood of ABH events in the pediatric population is also consistent with what was reported by Wheatley et al. (2018). While these findings may suggest the safe usage of NSAIDs for fracture pain management among pediatric patients, the analysis is limited, given that only two studies met the inclusion criteria. Further research should be completed on this topic in the pediatric population.
This study’s findings are also consistent with the meta-analysis reported by Al Farii et al. (2021), which exclusively included RCTs that examined the NSAIDs’ effects on fracture healing.19 Two of the included studies overlapped with the analysis in this study, while ten additional studies were included in the analysis in this study. While Al Farii et al. (2021) only included RCTs, the analysis in this study included all study types, allowing a larger number of studies to be included in the analysis. A relatively large Odds Ratio of 3.47 (95% CI: 1.68-7.13) was reported compared to the main analysis in this study, which reports an Odds Ratio of 2.10 (95% CI: 1.41 - 3.15).19 However, both studies are consistent in reporting an increased risk of ABH events with NSAID exposure post-fracture. The sub-analysis stratified by adult and pediatric populations in this study further contributes to the literature on the influence of NSAIDs on post-fracture healing.
Overall, fractures in children are far less likely to result in nonunion. Mills et al. reported the risk of nonunion to be approximately one in 500 for boys under 14 years old and girls of all ages, which increases to one in 200 for boys between 15 and 19 years of age.41 Skeletal immaturity, which supports a low nonunion rate, may also overcome the impairment of bone healing associated with NSAID use. While meta-analyses that examine the effect of NSAIDs post-fracture healing among pediatric populations are limited, systematic reviews have been published on the subject. A systematic review published in 2018 that explored the impact of NSAIDs on bone healing, including fracture and arthrodesis healing, found that all five studies in the pediatric population did not show impairment in bone healing.42 The five studies comprised two distal limb fractures and three spinal fusion studies. A more recent systematic review, published in 2021, which focused on the pediatric population and included both fracture and arthrodesis healing, also concluded that the use of NSAIDs in skeletally immature patients is a safe and effective pain management option.43 The meta-analysis in this study supports these conclusions and furthers the understanding of the subject by delineating the potentially safe use of NSAIDs in pediatric fracture healing.
Post-fracture pain management will remain an essential component of fracture treatment. While NSAIDs have been identified as a promising alternative due to their efficacious analgesic effect with lesser addictive potential compared to opioids, the adverse effect of impairing post-fracture bone healing adds a complicating factor to consider when determining the risk and benefit of the medication.8–10 Current literature suggests that lower doses and shorter duration of NSAID administration may not impair bone healing,16 indicating the need for more specific guidelines on the type, dosage, and duration of NSAIDs to consider prescribing for post-fracture pain management. An animal study has also demonstrated that fracture site and bone type may also modify the association between NSAID use and ABH, where NSAID use impaired unstable shaft fracture healing but did not impair stable metaphyseal fracture healing.44 To develop such guidelines, future studies should aim to determine the dosages/durations, fracture characteristics, and demographic characteristics in which NSAID therapy may be safe to use in fracture healing.
The quality of studies included in the analysis was high, contributing to the strength of this study. Further, the cumulative sample size was large despite the limited number of included articles. However, there are several limitations to the presented meta-analysis. First, despite the large and wide systematic approach to reviewing the literature, there is a possibility some articles that met inclusion criteria were not included in the final analysis. This possibility arises from the imperfect process of database searching using keywords and the manual selection of articles. Second, as with all meta-analyses, the current analysis required reporting raw subject numbers with non-zero events in each category from each study. Several publications reporting the association between NSAID use and ABH did not meet this requirement and were excluded from the analysis. Further, the study criteria specified the inclusion of articles written in English, creating a possibility of missing articles relevant to the meta-analysis but published in another language. These factors limit the comprehensiveness of the current analysis. Third, publication bias is a concern when interpreting the results of meta-analyses. Positive results tend to be favored for acceptance by journals, which generates a possibility of overlooking the inclusion of unpublished data that show negative results. In effect, this may inflate the observed association reported in this study. Lastly, the included studies demonstrated a wide variation in the prevalence of the outcome of interest. The variation may be accounted for by effect modifiers such as bone type, drug dosing, surgery type, and patient demographics, which are not accounted for in this analysis due to the lack of data to stratify by these variables.
Conclusions
The current literature suggests that the use of NSAIDs after a fracture interferes with timely post-fracture bone healing, especially among adults. However, among pediatric patients, NSAID use after fracture does not appear to interfere with fracture healing. The current literature still does not provide clear guidelines on the safe use of NSAIDs that are dependent on the fractured bone type, specific population characteristics, dosing, and duration. Future research focused on developing specific guidelines for the safe and effective use of NSAIDs after fractures may provide more detailed information for clinicians to use to help build an ideal postoperative pain regimen for their patients.
Declaration of Conflicts of Interest
The authors do NOT have any potential conflicts of interest for this manuscript.
Declaration of Funding
The funding for this work was provided by Drexel University College of Medicine.
Declaration of Ethical Approval
Given the nature of the study being a meta-analysis, an Institutional Review Board review was not applicable.
Declaration of Informed Consent
Given the nature of the study being a meta-analysis, informed consent was not applicable. There is no information in the submitted abstract that can be used to identify patients.
Acknowledgment
The authors acknowledge Janis Masud-Paul at the Drexel College of Medicine for providing technical expertise and support during the literature search of this study.